Dose of Confusion
OPELOUSAS, La. — On a chilly Friday night in January 2003, Christina Hutto took her 5-month-old
daughter to Opelousas General Hospital, a brown building squatting near the
center of this small Louisiana town.
A chubby girl with bright blue eyes and blond hair, Brianna had suffered from a cold
and fever for several days. A nurse suggested Tylenol. He scribbled the dose on
a piece of paper: one teaspoon every four hours.
Within days, Brianna was lying comatose in a pediatric intensive care unit — her
life threatened not by a deadly virus or rare disease, but by an accidental
overdose of one of the nation’s most popular over-the-counter pain relievers.
Her liver had been destroyed by a toxic byproduct of the medicine that was
supposed to help her.
The Huttos were blindsided. Like many Americans, Christina and Eric Hutto had
trusted Tylenol, a brand synonymous with safety. Tylenol, as the advertisements
proclaimed, was the No. 1 doctor-recommended brand of pain reliever; the one
hospitals used most; the one used by moms decade after decade.
Yet Tylenol’s pediatric products had the potential for lethal confusion — and
this was no secret to federal regulators or McNeil Consumer Healthcare, the
division of Johnson & Johnson that manufactures the drug.
For
at least 15 years, until 2011, McNeil continued selling two versions of Tylenol for young
children, despite knowing that parents and even medical professionals mixed them
up, sometimes with serious consequences. And the Food and Drug Administration failed
to intervene.
The two types of pediatric Tylenol had a counterintuitive difference. Drop for
drop, the strength of Infants’ Tylenol far exceeded that of Children’s Tylenol.
In addition, the active ingredient in Tylenol, acetaminophen, has what the FDA deems
a narrow margin of safety. The drug is generally safe at recommended doses, but the difference between the dose that helps and the dose that can cause serious harm
is one of the smallest for any over-the-counter drug.
By confusing the pediatric products and administering too much of the infants’
version, parents could inadvertently overdose their children. Other manufacturers
also made two children’s products with different concentrations of
acetaminophen.
Between 2000 and 2009, the FDA received reports of 20 children dying from acetaminophen
toxicity – a figure the agency said likely “significantly underestimates”
the problem. Three deaths were tied directly to mix-ups involving the two
pediatric medicines. Such errors may have caused some of the other deaths, but
the agency has acknowledged that its data lacks sufficient detail to determine
the precise cause.
Similar gaps exist in data for non-fatal liver injuries. The FDA has estimated it may capture less than 1 percent of such cases. Still, one small study found that confusion
between the two pediatric products was the most common reason for overdoses
among kids with acetaminophen-related liver damage. A study conducted by McNeil found that about one
child a year on average was hospitalized because of mix-ups involving its
drugs.
Such tragic accidents are among the reasons that between 2001 and 2010, there were
about twice as many deaths annually associated with acetaminophen than with all
other over-the-counter pain relievers combined, according to data from the
American Association of Poison Control Centers. On average, more than 150 Americans
die accidentally each year from acetaminophen poisoning, most of them adults, Centers
for Disease Control and Prevention data shows. Tens of thousands more are
hospitalized for overdoses.
Over a span of decades, federal regulators have been slow to protect consumers in
the face of the drug’s rising toll, and McNeil has argued against warning
labels, dosing restrictions and other protective measures that would have affected
its flagship brand.
The company and the FDA said they have done what they can to reduce overdoses
through labeling changes, public education efforts and other measures.
McNeil
pressed the FDA for years to allow it to add dosing instructions for children under 2 to its pediatric products; the government-approved label said only to
ask a doctor how much to give kids of that age. The company presented mountains of data and made
repeated public pleas in arguing for the change.
For more than a decade, the FDA deliberated on the company’s petition but never took action.
The issue “requires careful consideration of myriad inter-related factors,” the
agency said in a statement, including that “limited safety and efficacy data” existed
for acetaminophen in children under two. Today, the agency said, it is
continuing an effort to write “user-friendly” instructions for pediatric
acetaminophen.
All along, there was another option. At any time, the FDA could have pushed McNeil and
other manufacturers to switch to a single pediatric concentration. Or McNeil
could have done so voluntarily, ensuring that no one could mix up the dose.
Neither did.
Having two products allowed the company to sell to different consumers —
parents of babies and toddlers on the one hand, and older children on the
other.
Under oath in a lawsuit brought by the Huttos, McNeil’s former medical director,
Anthony Temple, was asked how often mixing up the two pediatric products had
led to liver injury. Over a 30-year period, he said, “There are maybe a couple
of dozen, maybe a little more, where incidents of significant liver injury has
occurred, and there’s probably a handful of those cases that were fatal.”
“And
for 25 years you’ve elected to continue to offer Infants’ Tylenol in the
concentrated form that has led to the death of babies, correct?” he was asked.
“Yes,
we’ve continued to do it,” Temple testified.
The company viewed moving to a single concentration as “a second-best option,”
Temple said in an interview.
“It’s easy looking back to say maybe we should have made a fix,” he said. But he explained
that McNeil believed for years that the FDA was going to add dosing
instructions. During that period, he said, the benefit to children warranted
keeping the two versions on the market.

“One death is too many.”
PeterMax Miller, a pharmaceutical marketing expert at the University of Colorado Denver and a former drug company executive
PeterMax Miller, a pharmaceutical marketing ethicist at the University of Colorado in Denver
and a former executive at a Johnson & Johnson competitor, said he saw the
math — and the morality — differently. First, do no harm.
Asked if just one infant death should trigger a drug maker to pull a product, he
replied that it would be “more than a trigger, that is a huge cannon.”
“One death is too many,” he added. “I would not have had any hesitation at all about
yanking it off the shelf overnight. Everywhere. And Johnson & Johnson knows
how to do that.”
Two years ago, as federal regulators were considering pulling the product, and as
lawmakers in Louisiana were weighing their own solution to the problem, McNeil
and other companies voluntarily withdrew the stronger formulation of their
infant drugs from U.S. store shelves.
That came too late for the Huttos.
Both Infants’ and Children’s Tylenol were on the shelves when Christina and her
mother walked into the local Walmart after leaving the hospital in early 2003. They
purchased what they had previously given Brianna — a bottle of the more concentrated
Infant’s Tylenol.
They drove to their home in Krotz Springs, a tiny community of trailers and small
tin-roofed houses nestled against a levee on the dark brown Atchafalaya River.
It was here that Eric Hutto had fallen in love with Christina after they met at
the “kissing log” — a creosote-covered post over a ditch that was a
make-out spot in town.
They were young when they got married, only 17. With Christina pregnant, the couple
worked hard to make their home safe for Brianna. Before her birth, they tore up
the old carpet in their trailer to put in new, allergen-free carpet. They did
the same thing in the kitchen, replacing the worn, old flooring with new
linoleum. They furnished a nursery for her.


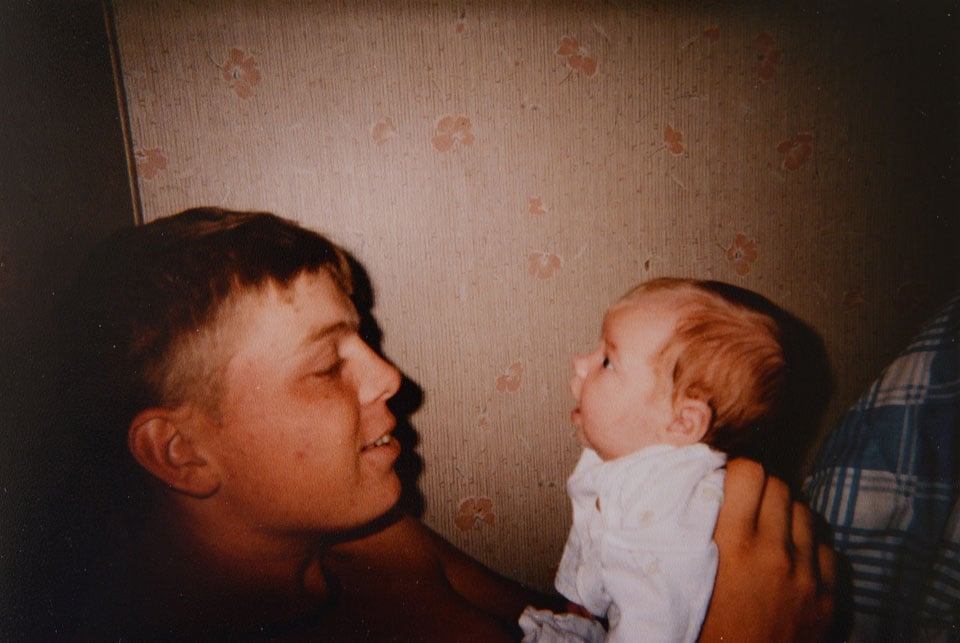
Christina loved her baby more than she could have believed — “like the sun had just opened a door in our lives,” she would say. Eric doted on his daughter, calling the day she was born the “happiest day.” (Photos courtesy of the Hutto family)
A construction worker who loved hunting and fishing, Eric doted on his young
daughter. He held her to him swaddled in a blanket, fed her, watched television
with her. Christina loved her baby more than she could have believed —
“like the sun had just opened a door in our lives,” she would say.
Over the weekend, the Huttos remembered, they dutifully began giving Brianna the
dosage recommended by the nurse at the hospital. They used a small plastic cup
with teaspoon indicators to measure the syrup. They used the dropper that came
with the package to administer it. They made sure they were giving her the
medicine at the right time intervals — no sooner than every four hours.

“There was no crying. There was no movement. She just became a vegetable.”
Christina Hutto, Brianna’s mother
By Sunday morning, Brianna seemed worse. She was restless and vomiting. By that
evening, she was listless. She wasn’t even responding to the sound of
Christina’s voice. “Sunday, there was no crying. There was no movement. She
just became a vegetable,” Christina said.
Christina had to go to her job at a truck stop, so Eric and his father, Jimmy, the local
fire chief, decided to take Brianna back to Opelousas General.
As the evening wore on, things went from bad to worse. Brianna wasn’t responding
to stimuli. She was dehydrated. The doctors were having trouble getting an IV
into her. Her veins kept collapsing. Christina rushed to the hospital. She and
Eric spent a sleepless night at the hospital with Brianna.
At the morning shift, Brianna’s regular pediatrician came into the hospital.
Christina remembers that she saw Brianna and immediately screamed: “Why wasn’t
I called? This baby is in bad shape!”
The doctor was confused. She suspected Brianna had ingested poison, but didn’t know
what. She asked Eric if the family had lead or other dangerous substances in
the house. Finally, she ordered up tests to check for liver damage and the
levels of acetaminophen, the active ingredient in Tylenol, in Brianna’s blood.
The
results showed Brianna’s liver enzymes were nearly 200 times higher than
normal. And the amount of acetaminophen in her blood indicated that she had
taken far more than the recommended dose.
Time was now of the essence. McNeil had helped to fund the development of an
antidote to acetaminophen poisoning that is remarkably effective if administered
within the first eight hours. Then its efficacy declines. Brianna had passed
the window. Her liver was failing fast. But there was still a chance.

“It was like it was just a tunnel we was riding through that never was going to end.”
Eric Hutto, Brianna’s father
A medical helicopter whisked Brianna to New Orleans Children’s Hospital, where
she could receive more specialized care.
There was no room on the helicopter for the Huttos. So Christina, Eric and Jimmy
piled into Jimmy’s pickup, and started driving down Interstate 10, past swamp
and cypress forest, toward New Orleans.
The two-hour drive was agony. Christina sobbed the entire way. Eric was bewildered.
Three days ago, he had a child with a fever. Now she was being airlifted for a
liver transplant.
“It was a long, long ride,” Eric said. “It was like it was just a tunnel we was
riding through that never was going to end.”
It would take several years to sort out what happened. Opelousas General, as it
turned out, did not use Infants’ Tylenol — precisely because hospital
administrators wanted to avoid the risk of overdosing by confusing two
different formulations. Nurses were used to giving dosage instructions based on
the less-potent form, Children’s Tylenol.
So when she first got to the hospital on that January night, Christina said, she told
the intake nurse that she had been giving Brianna Infants’ Tylenol. She said
she pulled the bottle out of the diaper bag to show the nurse.

Brianna was “very, very happy, very outgoing,” according to Christina, her mother. But that all changed when she got sick. (Photo courtesy of the Hutto family)
After Brianna was examined, a nurse in the emergency room handed Christina and her
mother a form, which recommended ¾ of a teaspoon of Tylenol. Christina’s
mother questioned the dose because she thought it was too high. When the form
came back, the nurse had increased the dosage to 1 teaspoon.
However, nowhere on the form that the nurse gave Christina did it say the teaspoon dose
was for Children’s Tylenol. And when Christina bought Infant’s Tylenol, as she
had before, there was no way for her to double-check the nurse’s instructions,
because there was no dosing information for a child under 2.
It was hardly a new problem.
McNeil
built its business partly on providing relief to sick children. When it began
selling Tylenol in 1955, one of its first products was a liquid form for
children. In 1957, McNeil introduced a second pediatric medicine: Infant’s
Tylenol. Originally prescription drugs, they became available as
over-the-counter products by 1959.
The infant’s formula, meant for kids under 3, contained more than three times as
much acetaminophen as the same volume of the children’s version, meant for
those up to 12. Though it might seem counterintuitive to make a stronger
Tylenol for younger kids, there were practical reasons to design the products
this way.
The more concentrated infant’s formulation meant that parents had to force fewer
drops down the throats of fussy babies to deliver the same amount of medicine. Having
two concentrations “was considered a rational strategy to facilitate dosing for
infants and children at the time it became available,” the FDA said in a
statement.
Some experts believe that isn’t persuasive. “Babies drink 4 to 8 ounces of formula
at a time. They can take a teaspoon of liquid,” said James Heubi,
a pediatric gastroenterologist at Cincinnati Children’s Hospital Medical
Center who has testified in court against McNeil. “There’s absolutely no reason
to have two different concentrations on the market.”
Having two products is a common sales strategy, helping to reach different
demographics. Johnson & Johnson does not release revenue information for
Tylenol products, so it isn’t publicly known how much the two pediatric formulations
generated. Industry-wide, infants’ strength products accounted for 12 percent
of sales of over-the-counter acetaminophen in 2009 and children’s strength products
accounted for about 16 percent, according to an FDA study.

“There’s absolutely no reason to have two different concentrations on the market.”
James Heubi, a pediatric gastroenterologist at Cincinnati Children’s Hospital Medical Center
McNeil did not directly answer why it continued to keep two concentrations on the
market. The company said that the “standard of care” in the U.S. included
providing more concentrated products to infants to facilitate giving the
medicine to babies.
“Sales were not an issue” in deciding whether to keep the two concentrations on
drugstore shelves, said Temple, the company’s retired medical director.
Understanding of acetaminophen’s potential to harm children began emerging decades ago.
By the late 1960s, science showed that, at certain doses, acetaminophen could
damage or even destroy the liver. Some research found that children were less
vulnerable than adults to the drug’s toxic effects, but in a 1975 article, Barry Rumack, a
toxicologist whose work McNeil later funded, noted the death in 1970 of a 3-year-old in England from acetaminophen poisoning. (The drug is known there and in many other countries as paracetamol.)
For many years, the chief concern was “unsupervised ingestion” – children
getting into medicine bottles and taking too much acetaminophen. Such accidents
are the reason for the majority of emergency room visits by kids suffering from
potential overdose, studies show.
In those cases, parents often rush their child to the doctor, increasing the
chance the antidote will be administered in time for it to work. But when
parents or healthcare providers mixed up the two concentrations of pediatric
Tylenol, they often wouldn’t realize anything was wrong for days, until symptoms
became severe and the antidote was less likely to be effective.
In 1994, parents overdosed a 14-month-old girl by confusing the two Tylenol products.
She lived but underwent a liver transplant. In a later statement quoted by newspapers, McNeil called her case
a “tragic error and confusion on the part of the parents.”
Two-thirds of parents who brought children under 7 into city emergency rooms did not know
the difference between concentrated infant’s drops and other formulations of acetaminophen,
a study
published in 2000 found.
As McNeil saw it, the problem had always been the label and its lack of
instructions for children under 2. The FDA’s rationale was that parents with children
that young should speak to a doctor in case the symptoms indicated a more serious
condition.
In
1983, McNeil’s Temple tried to address this issue by writing a scientific
article that included dosing instructions for children by weight and age. The
company began handing out tear sheets with the information to doctors to pass on to parents. The tear sheets also functioned as a marketing tool for Tylenol, according
to court records, offering discounts for the medicine.
Temple, whose research was supported by McNeil, later testified that the company considered
the tear sheets an imperfect solution.
Starting in the mid-1980s Temple said, McNeil officials publicly and privately pushed
the FDA to act — without success.
As research piled up in the 1990s on pediatric deaths and injuries, the FDA held a
series of public meetings at which McNeil and others advocated for adding
dosing instructions.
Doctors and pharmacists expressed growing concern about the two children’s
concentrations. In 1997, Dr. Cheston Berlin, an influential professor of
pediatrics at the Penn State University College of Medicine, said pediatricians
were “mystified” by the different formulations and urged drug makers to move to
a single version.
McNeil didn’t take this step. Nor did the FDA require it.
At the same hearing at which Berlin testified, an FDA advisory committee
recommended adding dosing information for children under 2 to acetaminophen
labels.
In 1999, the FDA approved such dosing instructions for pediatric medicines containing
ibuprofen, another over-the-counter pain reliever, including Motrin, a brand marketed
by McNeil.
But for products with acetaminophen, the FDA spurned the advisory committee’s
recommendation. The agency insisted that the label should continue to tell
parents to ask their pediatricians for the appropriate dose.
McNeil undertook other efforts to combat pediatric overdose. The company designed a special
safety cap that made it more difficult to pour out the more concentrated
infants’ formula.
The company also filed a formal petition asking the FDA to allow dosing instructions for young kids. It marshaled a growing stack of evidence that
dosing confusion persisted even among consumers who sought medical advice.
More than half the calls to McNeil’s consumer hotline concerned parents with
children under 2 asking for dosing information, McNeil told the FDA. Moreover,
a company analysis showed, most dosing errors occurred when parents had
obtained instructions from health professionals.
The FDA still put off action, saying that acetaminophen makers needed to supply even
more data showing how the drug affected kids of different weights and ages.
Unlike ibuprofen, acetaminophen had been approved under older safety rules
which did not require extensive testing. And, the agency said in a statement, “there
was not a lot of data on which to base dosing” for children using
acetaminophen.
Dr. Charles Ganley, an FDA official who was in charge of regulating
over-the-counter drugs, said at a 2002 hearing that the agency was “struggling”
with McNeil’s request for dosing instructions. The agency had even written a
proposal for instructions, he said, but there were complex issues involved —
including the agency’s own burdensome rule-making procedures.
“It’s
just not as straightforward as folks think,” he said at the hearing. “But it is
a priority to get done.”
Still,
more time passed with no decision — and no guidance for parents like the
Huttos.
–>
As soon as Christina and Eric got to New Orleans Children’s Hospital, they rushed
up to see Brianna.
They were shocked at what they saw: Brianna was on a hospital bed, tubes running
everywhere. She seemed to be swelling up, almost like a balloon. Her skin was
tight, her lips were cracked. Beeping noises filled the room.
Christina called out to her, and Brianna briefly opened her eyes. She raised her arms
toward her mother and let out a strange cry. Christina rushed over and started
singing a lullaby. Suddenly, Brianna started spitting up. Christina broke.

About two weeks after her first Christmas, Brianna died from acetaminophen toxicity. (Photo courtesy of the Hutto family)
“I couldn’t take it anymore. I ran out of the room. I couldn’t take it,” she said.
The doctors told the Huttos that Brianna was in an advanced stage of acetaminophen
poisoning. Her liver was dying, and gases were building up inside her. The only
hope was a liver transplant. By chance, the University of Nebraska, which had a
specialized liver transplant center, had surgeons available for an operation.
A day after they arrived in New Orleans, the Huttos were on the move again, flying
in a medical jet to Omaha.
Before they left, they had a priest administer last rites. Brianna had slipped into a
coma. In the ambulance on the way to the hospital, her heart stopped beating.
The nurse resuscitated her. Doctors told the Huttos that Brianna would have to
breathe on her own in order to be considered a candidate for a transplant.
“I kept saying, “Hold on, hold on, hold on,’ ” Christina said. “She was fighting
so hard.”
When the Huttos arrived in Omaha, doctors rushed Brianna into an intensive care
unit. The Huttos had their livers tested for compatibility with Brianna.
Christina could have part of her liver removed to transplant into Brianna.
That morning, Eric bought a stuffed animal for Brianna from the hospital store, a
small purple lamb with the word “Faith” embroidered on its neck. As he returned,
he saw the doctors rushing in and out of Brianna’s room.
“Something’s going on with Brianna,” he told Christina. A moment later, a priest entered the
room, a team of doctors and nurses behind him. Brianna’s kidneys, heart and
liver had shut down.
Brianna Hutto died at 11 a.m., Jan. 8, 2003.

“When you pick out colors, it’s supposed to be for prom dresses or Christmas pictures, not colors of the lining of her casket.”
Christina Hutto, Brianna’s mother
Christina had been on the phone with her mother when the priest walked in. She screamed.
She dropped to the floor. Eric just stood there; he couldn’t comprehend what
was happening.
“I felt dead. I felt empty inside, like my world ended,” he said. “It’s just the
worst feeling imaginable. You can’t explain it besides my heart stopped.
Everything stood still.”
The next few days passed in a blur. The autopsy results showed the cause of death: liver
failure secondary to acetaminophen toxicity.
The couple flew back to New Orleans, Brianna’s body in the plane’s baggage
compartment. They arranged for a viewing and funeral services at the church in
Krotz Springs.
“You know, when you pick out colors, it’s supposed to be for prom dresses or
Christmas pictures, not colors of the lining of her casket,” Christina said.
Brianna was buried in a small pink casket, in a grave behind the church.
Nearly a year passed before the Huttos decided that no other parents should suffer as
they had.
On Jan. 7, 2004, one day short of the anniversary of Brianna’s death, the two
teenagers filed a lawsuit against Opelousas General Hospital and McNeil, whose
parent company, Johnson & Johnson, was one of the biggest corporations in
America.
Their claim against McNeil: The drug company had not disclosed the full dangers of
its product.
Have you or someone you know experienced acetaminophen poisoning – by accident or on purpose?
About 160 Americans die accidentally each year from acetaminophen poisoning — and about the same number use the drug to commit suicides each year. ProPublica is seeking stories of those who have been harmed.
Six years of legal wrangling took place before the lawsuit finally went to trial on
June 15, 2010, in a courtroom in the basement of the old stone courthouse that
dominates the Opelousas town square.
Before trial, Opelousas General Hospital and the attending physician admitted
responsibility for giving the Huttos the wrong dosing information. They settled
with the couple, leaving McNeil as the sole defendant.
McNeil’s defense was simple. The label on Infant’s Tylenol said to call a doctor for
dosing instructions for a child under 2. The label also warned that “serious
health consequences” could result from not following instructions —
language approved by the FDA. The hospital had given out bad advice. Tragic as
the case was, the couple had exceeded the recommended dose of Tylenol. McNeil
was not to blame.
McNeil brought a large team of experienced lawyers to represent it at trial, but the
company was not used to facing a jury. Of more than 150 lawsuits involving
Tylenol examined by ProPublica, only a handful went to trial. Most were settled
out of court, the terms sealed as part of the deal.
The Hutto trial, which lasted six days, featured emotional testimony from Christina
and Eric. At one point, Christina broke down on the stand.
For McNeil, Temple testified that acetaminophen was the safest pain reliever on the
market. He acknowledged that the company was aware of the problem of parents
confusing Infant’s and Children’s Tylenol, but blamed the FDA for preventing
the company from putting dosing instructions on the label.
An attorney representing Opelousas General Hospital pressed Temple on why McNeil
hadn’t just eliminated one of the products if it knew that parents were making
dosing errors and that children were being harmed.
“Rather
than pull the products off the shelf, that’s sort of the cost of doing
business?” the lawyer asked.
“McNeil felt that the benefit as — really it’s the FDA because it hasn’t pulled
it off either — but the benefit to children of having the product
available was appropriate to keep it in the marketplace, yes,” Temple
responded.
The Huttos’ lawyers painted the trial as a David and Goliath affair, McNeil versus the
young Louisiana couple.
The jury, which wasn’t told that the hospital had settled, wound up splitting the
blame. The hospital was 70 percent at fault. McNeil was 23 percent at fault.
Christina and her mother were 7 percent at fault. The jury awarded total
damages of $5 million. Proportionally, the decision meant that McNeil owed the
Huttos a little more than $1 million. But under a Louisiana law that limited
damages paid by the hospital, the Huttos wound up with a little more than $2
million, before factoring in lawyers’ fees and trial costs.
Over the next two years, the company appealed the verdict to the U.S. Supreme Court.
In fall 2012, the court declined to hear the case, letting the original jury
verdict stand.

“Our hearts go out to those who have suffered from acetaminophen overdose, and to the families of those who have lost their lives as a result.”
Statement by McNeil Consumer Healthcare, the Johnson & Johnson unit that makes Tylenol
The company’s legal tactics left the Huttos bruised and stunned. One McNeil
attorney had told them that he was sorry for their loss. But the fight left
them convinced that McNeil cared more about money than the safety of its
medicine.
“They probably spent more on lawyers than they did on the money they paid us,” Eric
said. “Anybody with a heart, whether you own a business or not, should have
said, ‘We’re at fault, proven guilty, here you all go. I hope y’all’s life gets
better.’”
McNeil did not respond to questions regarding the Hutto case, but offered condolences
to people who had suffered harm from the drug.
“Our hearts go out to those who have suffered from acetaminophen overdose, and to
the families of those who have lost their lives as a result,” the company said
in a written statement. “We will continue to work hard to educate and warn
consumers of the dangers of acetaminophen overdose, reminding them to read
labels on all medicines before taking them, to take medicines only as directed
and to be aware that any medicine they take has risks.”
Christina did not find the company’s statement in any way consoling. She had read the
label, followed its directions and lost her child.
“I did all those things,” she said.
As the Huttos’ case inched toward a conclusion, McNeil’s decision to continue
selling two kinds of pediatric Tylenol with different strengths began to draw
fire from health experts and regulators.
In 2009, the FDA convened a panel of experts to consider restrictions on
acetaminophen, including whether to require companies to produce only one
concentration of the drug for children. The FDA presented evidence that 10
children under 6 had died from acetaminophen poisoning between 2000 and 2009
— including Brianna Hutto.
Dr. Edwin Kuffner, McNeil’s vice president for medical affairs, and Temple both
argued at the hearing that the answer was clear dosing instructions.
“It’s
the labeling, not the concentration,” said Temple, who had retired from McNeil
but flew in for the hearing at his own expense. He called the FDA’s inaction on
the matter “astounding,” noting that the
agency had promised to issue a ruling every year between 2003 and 2008 and never
did.
The panel didn’t vote on whether to add dosing instructions. It voted, 36-1, that
the FDA should mandate a single concentration of acetaminophen for children.
“I’ve
not heard, in the information that we’ve reviewed, any compelling reason for why
there should be these two formulations,” said one panelist, William Cooper, a
professor of pediatrics at Vanderbilt University.
Once again, however, the FDA took no action on its experts’ recommendation.
Just one month after the meeting, McNeil wrote to FDA officials, proposing a plan in
which it and other manufacturers would limit pediatric syrups “to a single
concentration.” But it took almost two more years for McNeil and its
competitors to take that step.
In April 2011, the Huttos’ attorney persuaded a Louisiana state legislator to introduce
“Brianna’s Bill.” The measure said only pharmacies could sell children’s and
infant’s acetaminophen products and that parents had to get written
instructions when buying them.
In May, the FDA scheduled yet another hearing at which an expert panel examined
the safety of children’s acetaminophen. Two weeks before the meeting, McNeil
and other makers of acetaminophen announced what they called a voluntary
decision to eliminate the two different concentrations of kids’ products.
Same Teaspoon, Different Dose
5mL of Infant’s Concentrated Tylenol has more than three times as much acetaminophen as 5mL of the Children’s version.

In a letter to physicians, McNeil said the company
believed a single concentration would improve patient safety. “The purpose of
this transition is to help minimize the potential for medication errors due to
confusion between infants’ and children’s acetaminophen products having
different concentrations,” Kuffner wrote.
At the hearing, the FDA laid out its best estimate of the human cost of pediatric
overdoses. The agency updated its analysis from 2009, finding that twice as
many children — 20 — had died due to medication errors involving
acetaminophen as it had reported before.
Moreover, agency officials said, they had come to suspect there were vastly more
pediatric injuries from acetaminophen toxicity than they had known.
FDA researchers had compared two databases: the agency’s own MedWatch database, to
which pharmaceutical companies are supposed to report serious drug side
effects, and that of the American Association of Poison Control Centers, which
collects data voluntarily submitted on poisoning cases across the country. The FDA
estimated that the poison control center data captured about 400 pediatric
liver injuries related to acetaminophen in 2008 and 2009 that its own reporting
system did not.
Perhaps most striking was an analysis presented by Randall
Bond, a pediatrician and poison control center director who testified on behalf
of the Consumer Health Products Association, a trade group including McNeil and
other acetaminophen makers.
From
2000 to 2004, Bond’s research showed, moving to a single concentration of
acetaminophen for pediatric syrups could have prevented six serious injuries
and two deaths.
Brahm Goldstein, a nonvoting member of the advisory panel who represented the
pharmaceutical industry, criticized McNeil and other acetaminophen makers for
not acting sooner.
“I’d like to congratulate the industry colleagues on moving to single dosing, but I
would also like to chastise them,” Goldstein, an executive at Ikaria, a
pharmaceutical company focused on critical care, told the group.
“I don’t know the specific number of children who have been injured and/or died
because of medication errors due to this specific problem, but the fact that it’s
taken over 10 years to get where we seem to be going this summer and later this
year I think is shameful.”
The panel voted unanimously to recommend that the FDA add dosing instructions for
children under 2 on pediatric acetaminophen.
To this day, the FDA has not put rules in place to require a single concentration
of acetaminophen for children, relying instead on the industry’s voluntary step
to move to a single concentration. This August, the industry filed a new petition requesting permission to put dosing instruction for kids on the single ingredient product remaining on the market.
In a written response to questions for this article, the agency said it was
continuing to work on a formal regulation to settle pediatric dosing once and
for all.
“We are working to reconcile the many related factors and establish a user friendly
approach to proper pediatric dosing by age and weight in a future rule for
children’s acetaminophen,” the agency said.
Overseas,
in markets such as Mexico and Canada, McNeil still sells Infants’ and Children’s
Tylenol with different concentrations of acetaminophen. The labels in those
places give dosing information for children under 2.
The Huttos fell apart after Brianna’s death.
Christina’s grades plummeted, and she dropped out of college. She blamed herself for
Brianna’s death, for not doing a better job of protecting her.
She divorced Eric. “I couldn’t stand being with someone, looking at that person
knowing every day what I didn’t have anymore,” Christina said. “So I filed for
divorce and just lost it. I went crazy.”

“I can’t tell you how many times I wake up in the middle of the night to make sure they’re still breathing.”
Christina Hutto, Brianna’s mother
Christina moved out of Louisiana, determined never to go back. She has remarried and now
has four children.
“I can’t tell you about the fear that I have of my children getting sick. I can’t
tell you how many times I wake up in the middle of the night to make sure
they’re still breathing,” she said. “And I will do this for the rest of my
life.”
Eric plunged into depression.
“Thoughts of suicide was there,” he said. “How I didn’t do it, I don’t know.”
Today, Eric lives in a small town on a spit of land south of Baton Rouge. He used the
money from the settlement to start an auto upholstery business. He hunts. He
fishes. And he visits Brianna’s grave once or twice a month.
“I tell her I love her and I miss her and wish she was still here and to watch
over us and stuff,” he said. “I try to go by when I can. Sometimes I just
don’t. It doesn’t feel good when you do go. It’s a sad, empty feeling.”
Related Story:Use Only as Directed
What We’re Watching
During Donald Trump’s second presidency, ProPublica will focus on the areas most in need of scrutiny. Here are some of the issues our reporters will be watching — and how to get in touch with them securely.
Learn more about our reporting team. We will continue to share our areas of interest as the news develops.

Sharon Lerner
I cover health and the environment and the agencies that govern them, including the Environmental Protection Agency.

Andy Kroll
I cover justice and the rule of law, including the Justice Department, U.S. attorneys and the courts.

Jesse Coburn
I cover housing and transportation, including the companies working in those fields and the regulators overseeing them.
If you don’t have a specific tip or story in mind, we could still use your help. Sign up to be a member of our federal worker source network to stay in touch.











What Do You Think?
Should American drugmakers have to limit acetaminophen pills sold over-the-counter?
Should the FDA require all acetaminophen drugs to carry black box warnings?
Who’s most to blame for deaths caused by mixing up Tylenol projects for infants and children?