In 2021, after Philips Respironics sold millions of defective medical devices to those who struggle to breathe, the federal agency charged with protecting the health of the American public swept in.
The Food and Drug Administration accused the global powerhouse of a succession of mistakes — casting aside test results and health risks — long after the company discovered an industrial foam embedded in its breathing machines could break down and send tiny particles and fumes into the lungs of patients.
The FDA maintains that it acted as soon as it learned of the safety concerns in April 2021, just weeks before Philips launched one of the largest recalls of its kind.
But a ProPublica and Pittsburgh Post-Gazette investigation found that in the years leading to the recall, the FDA repeatedly failed one of its most critical missions: alerting the public about devices that can inflict serious harm. Over the course of a decade, the agency missed a pattern of warnings from health care workers, patients and others that something was very wrong with the company’s popular sleep apnea devices and ventilators.
From 2011 to 2021, Philips sent hundreds of complaints about the machines to the FDA, none of which resulted in alerts to doctors or patients. One report described a “black powder substance” inside a ventilator. Another noted foam that was “loose and tangled.”
Scores specifically cited “contamination,” a red flag that experts say should have prompted an immediate inquiry because the machines send air directly into the noses and mouths of users, including infants and the elderly. It is unclear who, if anyone, read the reports at the agency.
After the recall, the FDA said that Philips had held back thousands of additional complaints, compromising a public warning system meant to inform consumers about life-threatening device failures. The FDA allowed the company to submit reports years later, so far without penalty.
The FDA also permitted Philips to submit follow-up reports about those late complaints and update the dates that the company first received them. Then the agency concealed the original dates from the public, obscuring how long Philips had the warnings in hand before turning them over to the government.
“That’s regulatory failure,” said Paul Pelletier, a former federal prosecutor who once led health care probes for the Justice Department. “There is no other way to say it. They dropped the ball.”
ProPublica and the Post-Gazette spent more than a year investigating the Philips CPAP recall and the FDA’s response, analyzing 17 million reports in a complaint-tracking system open to the public that has long served as the backbone of the government’s oversight of medical devices.
Created about three decades ago to detect repeated breakdowns, the system relies on companies whose profits are tied to the success of their products to quickly disclose problems and on the FDA to review the warnings, respond to them and hold device makers accountable when they delay reporting on time or fail to do so at all.
Federal law has long required manufacturers to disclose malfunctions, patient injuries and deaths within 30 days. But since 2010, Philips and other subsidiaries of Dutch parent Royal Philips have been late in submitting at least 60,000 complaints to the FDA — often by years, government records show.
That includes more than 3,700 complaints about the recalled continuous positive airway pressure, or CPAP, machines and ventilators, which a ProPublica and Post-Gazette story reported in September. The delays came as patients using the machines suffered from inexplicable respiratory infections, cancers, liver and kidney problems, and other illnesses — ailments that some medical experts fear are tied to the crumbling foam.
Philips said early on the devices could send potentially “toxic and carcinogenic” material into the masks of patients, and the FDA classified the recall as the most serious, for defects capable of causing severe injury or death.
Philips has a history of withholding complaints about medical devices it manufactures. The FDA’s own inspectors have previously cited the company for failing to turn over reports about safety breakdowns involving widely used CT scanners and defibrillators, public records show. Though the agency can pursue criminal charges for the delays, Philips has never faced such penalties.
The lapses in enforcement have not been limited to Philips.
In analyzing complaints in the government’s tracking system, the news organizations found that other leading device makers have submitted hundreds of thousands of late reports to the FDA, in some cases waiting years before disclosing the information. Last year alone, 1 in 8 reports — more than 232,000 complaints — were submitted past the 30-day deadline, leaving regulators and the public without badly needed safety information.
The FDA has sent warning letters to errant companies, but criminal charges are rare.
The analysis also exposed the troubling use of the FDA directive instructing manufacturers to update the dates they became aware of potential defects when providing follow-up reports to the agency.
A review of more than 100,000 complaints submitted since 2013 by two dozen large medical device makers showed that dates were changed on 1 in 5 reports, including those about flawed pacemakers, prosthetics, dialysis machines, and even screws and plates for bones.

The news organizations found the discrepancies in the dates after obtaining the original reports from a private company that stores FDA data and comparing them to the reports as they appear in the agency’s public database.
Medical experts and lawyers who rely on an accurate timeline of breakdowns say that permitting companies to submit late reports or change dates undercuts the nation’s primary system for tracking the safety of medical devices.
“I never imagined that this would be allowed,” said Madris Kinard, a former FDA analyst who was brought in to fix the system in 2010 and quit four years later. “It boggles the mind.”
In a statement, the agency acknowledged that device makers are directed to replace original dates when updating reports in the system but said copies of earlier versions — not available in the public tracking system — are kept separately at the agency.
The FDA did not respond to questions about why such a policy is in place but said that the tracking system is “just one source of information” about faulty medical devices.

The agency defended its handling of the Philips recall, saying officials continue to “take steps to protect the health and safety of individuals using these devices.”
The FDA said it received complaints about “general contamination issues” before the recall but that the debris could have been caused by external sources unrelated to degrading foam. Complaints that specifically described problems with the foam did not indicate that any patients had been harmed, the agency said. The FDA said all complaints are read but did not specify when it reviewed them.
More than two years after the recall, Philips maintains that patient safety is a top priority and its machines are unlikely to cause “appreciable harm.”
In a statement, the company said that it turned over the late complaints about its breathing machines to the FDA “out of an abundance of caution” and that it did not initially believe the complaints needed to be reported to the government. The company did not say why so many other complaints were reported late. It said the date changes were “consistent with regulatory guidelines.”
Philips and Philips Respironics, the company said, “share the same objectives as the FDA.”
Philips has disclosed that it is in discussions with the agency about a consent decree that could compel the company to make significant improvements. The FDA said it could not comment on potential enforcement action.
Criticism of the agency’s oversight of the industry is not new.
Over the years, the FDA has promised to overhaul the way it detects dangerous medical devices by relying more on real-time data in medical registries, electronic health records, insurance claims and other sources. Under Jeff Shuren, a neurologist and attorney who has long led the FDA unit that regulates devices, the agency has directed millions of dollars to that effort.
But to carry out the plan, the FDA in 2016 turned to an organization whose members include major device makers, such as Philips. The group paid for conferences, consultants, travel and pay for its executives, public records show.

Years later, the promised system is still not in place.
“It’s very disappointing that we continue to see delays in public notifications of serious device safety issues from the FDA,” said Dr. Rita Redberg, a cardiologist and expert on medical device safety. “We learn about [problems] after years and years and lots of preventable injuries and deaths. It’s such an avoidable disaster.”
“I Was Just Horrified”
In late 2012, hospitals in three U.S. cities scrambled to contain a disturbing pattern of infections among patients who had undergone exams for digestive illnesses.
Eventually, investigators found the connection: a hollow, lighted tube made primarily by medical device maker Olympus that doctors snake down the throat and stomach to peer at the small intestine.
By the time the maker of the duodenoscope launched a recall about three years later, the FDA had received dozens of reports about deaths, infections and injuries to the bowels and other organs, the ProPublica and the Post-Gazette analysis found.
A Senate investigation in 2016 faulted the FDA and its “outmoded” complaint-tracking system for allowing the crisis to continue well after the devices started sickening patients with virulent infections.
“Preventable tragedies,” a Senate report called the crisis after doctors around the world raced to treat patients. “A vivid example of the failure of FDA’s current system for tracking and monitoring the safety of medical devices on the market.”
The congressional probe came six years after an inspector general’s report found the FDA had allowed warnings about medical devices to sit untouched for weeks or longer.
At the time, about 20 people were assigned to read the complaints, which were kept in an electronic tracking system from the 1990s that couldn’t easily retrieve large numbers of related records or run comprehensive searches for key information. Reviewers often jotted down details about the most alarming cases on Post-it notes and tacked them to their computers.
“A huge number of reports weren’t read,” said Kinard, the former FDA analyst. “I was just horrified.”

As the complaints languished, thousands more came in. The increase was driven partly by the sheer number of products on the market, the vast majority approved through an expedited review process created in the 1970s and championed for years by the industry.
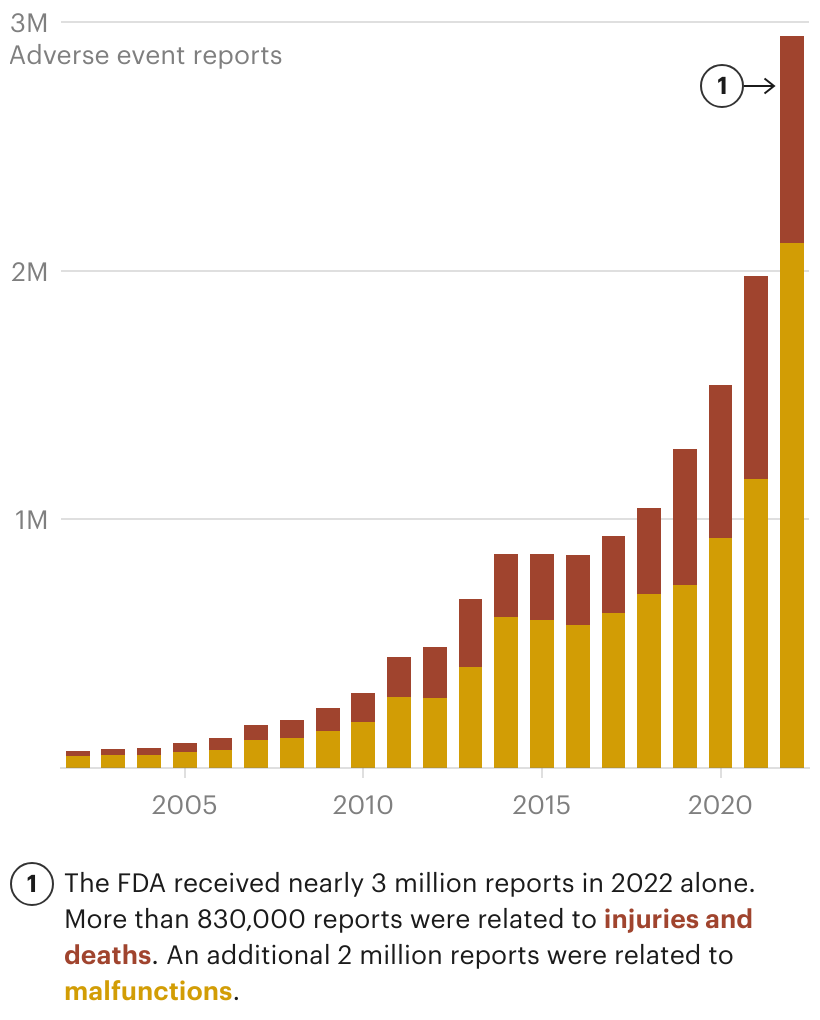
Last year, the FDA received 3 million reports about potentially defective devices — nearly 30 times more than in 2005, government records show. Nearly one-third described injuries and deaths.
The FDA, which regulates more than 200,000 types of medical devices, did not say how many people are currently assigned to screening the reports.
Reports About Medical Device Defects Skyrocketed in Recent Years
Manufacturers are required under federal law to turn over to the FDA reports of patient deaths, injuries and malfunctions that have the potential to cause harm.
Reports About Medical Device Defects Skyrocketed in Recent Years
Manufacturers are required under federal law to turn over to the FDA reports of patient deaths, injuries and malfunctions that have the potential to cause harm.
Amid concerns about the agency’s response time, the inspector general in 2009 faulted the FDA for not cracking down on companies that submitted late reports. In response, the agency pledged to offer “educational assistance” to manufacturers and conduct inspections for chronic offenders.
But years later, device makers have continued to turn over complaints months or years after they came in, ProPublica and the Post-Gazette found.
The FDA has significant power to address defective products or companies that ignore its rules by seeking criminal charges, fines and injunctions.
Olympus pleaded guilty and agreed to pay tens of millions of dollars in 2018 for holding back reports that would have exposed the scale of the problem with its duodenoscopes. Late last year, the company was warned again by the FDA about failing to disclose complaints on time.
Olympus acknowledged that it filed some complaints about duodenoscopes late but said the “disclosure failures” were not tied to patient injuries. The company said it is launching a new global complaints system to address the FDA’s more recent concerns.
“Olympus takes the FDA findings and feedback very seriously,” Olympus said in a statement.
Other companies have escaped penalty.
Device maker Becton, Dickinson and Company did not submit 25,000 reports dating back to 2010 about its defective infusion pumps until the devices were recalled nine years later, government records show. The pumps, linked to scores of injuries and at least one death, were malfunctioning while delivering medication and blood to critically ill patients.
In a statement, the company said that it turned over the late reports after the FDA carried out an inspection in 2020 and that none of the cases involved patient injuries or deaths. The FDA said it took steps to provide information to the public and work with the company.
Public health advocates and patients who have been harmed by defective devices, however, say the FDA too often fails to hold companies accountable.
Tess Schulman, a paralegal in North Carolina, struggled with rashes that her doctor said were caused by Essure, a contraceptive device manufactured by Bayer that was blamed for lost pregnancies and deaths and later pulled from the market.
“Why are we still allowing this to happen?” she asked. “Everybody thinks they are there to protect the public and they would not allow companies to sell something that wasn’t safe. We have a false sense of security.”
In a statement, Bayer said it continues to “stand behind Essure’s safety.”
Beyond the late filings, Olympus updated the dates on follow-up reports submitted to the FDA — more than 2,000 times in the case of its troubled duodenoscope, government records show. In each case, the change made it appear in the FDA’s tracking system as if the company had more recently received warnings when they had actually come in months or years earlier.
Like Philips, the company said it was following instructions from the FDA.
The directive has created vast inconsistencies in a system meant to inform and protect the public. ProPublica and the Post-Gazette found that other companies have often left the original dates intact, despite the FDA’s guidance.
The agency said it has the capability to root out “systemic” reporting problems, but experts say they fear the date changes serve companies seeking to conceal potential violations of the law.
“The risk is obvious,” said Michael Gonzalez, an Ohio lawyer who advises companies on health care compliance. “You don’t take what might be evidence in a case — and even your own culpability — and then alter or change it.”
Kinard, the former FDA analyst, said she discovered about a year ago that the agency had created a pathway for manufacturers to make the changes.
“It is an error on the FDA’s side that is being exploited by manufacturers,” said Kinard, who added that she had no idea the changes were so extensive. “I want to know … who has been taking advantage?”
An Unfolding Crisis
The steady series of reports about contaminated CPAPs and ventilators streaming into the FDA in the years before the Philips recall should have come as no surprise to the government.
The FDA had co-hosted a meeting with the nonprofit Association for the Advancement of Medical Instrumentation about ventilator safety in 2014, raising alarms about how material packed in the devices could contaminate the air quality and send “substances into the patient airway and lungs.”
When Philips finally announced a recall in June 2021, acknowledging the foam fitted in its machines could break down in heat and humidity, the FDA released a series of updates on its website but did not address the warning it had issued years earlier.

The agency also said little about the reports it had been receiving from Philips all along.
One of the first arrived in 2011, describing “black substance in the air path” of a ventilator, records show. Another the next year noted a “significant build up of dust and particulate.” Other reports were more detailed, describing problems with the foam itself. “Foam was found to be deteriorated,” read one report submitted to the FDA in 2020, about seven months before the recall.
It remains unclear whether anyone at the FDA at the time looked at the mounting evidence that something was amiss. The agency has since said that Philips submitted 30 reports between 2011 and April 2021 that specifically described foam degradation.
“We review and take seriously all reports of adverse events associated with medical devices and conduct additional evaluation and analysis when necessary,” the agency said. “We take prompt action and communicate publicly when appropriate.”
There is no evidence, however, that the FDA took any action as a result of the foam complaints or the hundreds of reports that described contamination.
While Philips forwarded some complaints to the government, ProPublica and the Post-Gazette reported that the company withheld thousands of others over a span of 11 years, including reports that described deaths among patients.
It wasn’t the first time Philips held back reports about malfunctioning medical devices.
In 2011, the FDA cited a Philips subsidiary for failing to turn over complaints about faulty imaging scanners, including at least two that reported the machines had caught fire, government records show. That same year, the agency found the company was withholding reports about emergency defibrillators that failed to work when patients with heart problems needed them.
A federal court eventually forced Philips to stop distributing defibrillators in the United States, but the order was lifted in 2020.
All the while, Philips was quietly scrambling to deal with the flurry of complaints about its popular breathing machines. And people were getting sick.
Eleven hundred miles away from Washington, in a trim white house in Baton Rouge, Louisiana, Richard Ieyoub rarely thought about the company behind the CPAP machine he had used for years.
The former attorney general of Louisiana, who helped lead a groundbreaking lawsuit against U.S. tobacco companies that ended in a massive settlement in 1998, was serving out a term as the state’s top oil and gas regulator.


He was also recovering from a rare form of mouth cancer; doctors had to remove a part of his jaw and then rebuild it during a 17-hour surgery to remove a tumor in 2017. The father of seven spent time in recovery at Jesuit retreats and at a family lake house, sitting by a fire pit and regaling his grandchildren with stories about parents, aunts and uncles who had emigrated from Lebanon and settled in rural Louisiana parishes.
After Philips launched the recall, Ieyoub, like the other CPAP machine users who had grown sick, said he began to question whether the device he had used for hours every night was to blame.
And as the prosecutor who took on the dangerous practices of tobacco companies, he wondered why the federal government did not warn the public years earlier.
“To think that so many people are going to suffer,” he said in an interview last year. “There has to be some kind of accountability.”
Early Warning System
Members of Congress have repeatedly questioned the FDA’s oversight of medical devices, especially in the aftermath of wrenching reports of injuries and deaths. Shuren, the top regulator, has long promised to keep the public safe.
“We will remain vigilant,” he said this year.
But the agency’s use of enforcement tools, including inspections and seizures, has dropped significantly in recent years even as the number of new devices hitting the market reached record levels.
The FDA said gaining entry to manufacturing plants during the COVID-19 pandemic was difficult. But the number of inspections started dropping in 2018, two years before the coronavirus crisis, and continued through last year, FDA data shows.
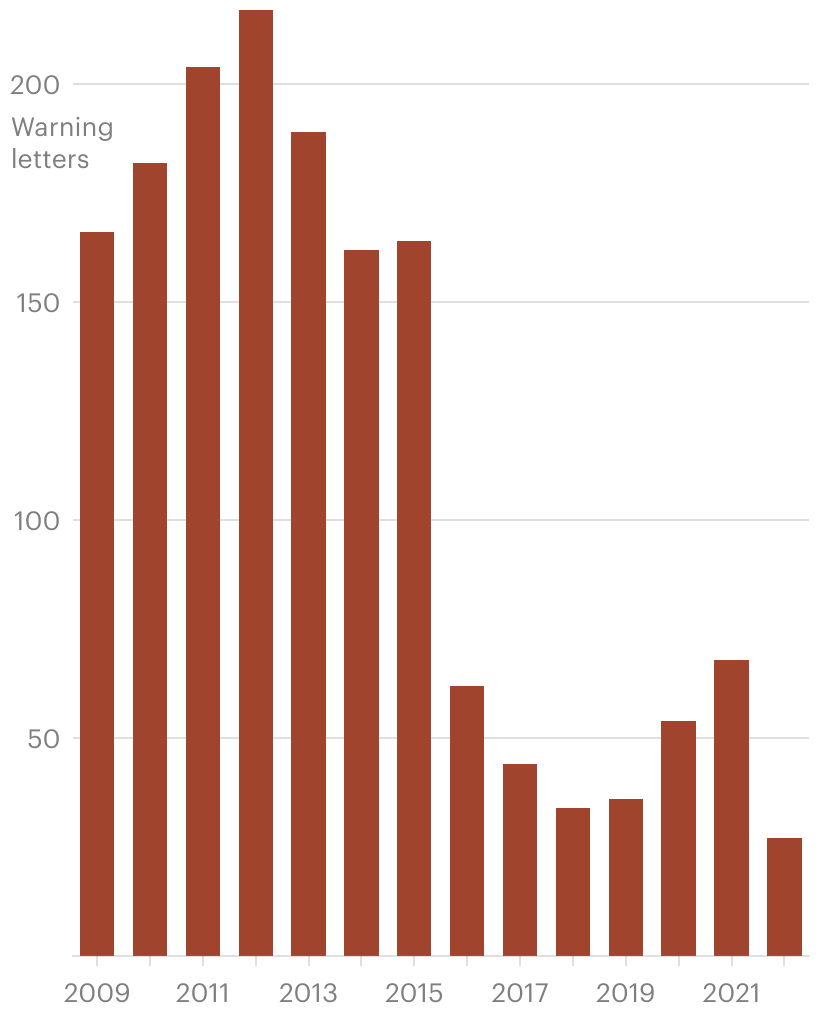
The number of warning letters, which the FDA considers the “principal means of achieving prompt voluntary compliance,” dwindled to 27 last year, down from 217 in 2012, records show. The use of injunctions and seizures against troubled device makers has also dropped.
The FDA Has Issued Far Fewer Warning Letters to Medical Device Manufacturers
Warning letters are used by the FDA to compel manufacturers to voluntarily comply with federal regulations. The agency has attributed the drop to difficulties gaining access to manufacturing plants during the COVID-19 pandemic.
The FDA Has Issued Far Fewer Warning Letters to Medical Device Manufacturers
Warning letters are used by the FDA to compel manufacturers to voluntarily comply with federal regulations. The agency has attributed the drop to difficulties gaining access to manufacturing plants during the COVID-19 pandemic.
Shortly after the congressional probe into the Olympus recall in 2016, Shuren and the FDA launched a bold plan.
That year, the agency awarded $3 million in seed money to the nonprofit Medical Device Innovation Consortium to establish a center that would bring together information from electronic medical records, insurance claims and medical registries.
Dubbed NEST, the National Evaluation System for health Technology, the initiative aimed to spur medical device innovation and advance an early warning system that would alert doctors, patients and regulators to device malfunctions actively occurring in medical settings.
Over the course of eight years, the FDA devoted millions of dollars to the effort. The nonprofit paid for travel, consultants, technology and bonuses, and about $400,000 a year in pay for its last executive director, records show. But the group has yet to develop a comprehensive new system.
Patient advocates and others have questioned whether Shuren — one of the most influential voices in the $185-billion-a-year U.S. medical device industry — pushed hard enough to see the plan succeed.
Shuren was the vice chairman of the membership committee at MDIC and has been a board member for years, records show.
One top FDA official said Shuren’s connection to the group, given the deep involvement of industry, has signaled to device makers that they have an ally in the agency responsible for regulating them.
“It smells to high heaven as far as I’m concerned,” said the official, who spoke on the condition of anonymity because he was not authorized to comment publicly.
The FDA said Shuren adheres to all ethics and conflict of interest guidelines. Shuren declined an interview request and to answer written questions.
The FDA noted that representatives from other government agencies are also on the board of MDIC and that a network of hospitals, medical centers, clinics and practitioners is bringing together data about devices. The agency said it requires funding to go directly to building and maintaining the network of partners.
In a statement, MDIC said that NEST, a “sub-group” within the nonprofit, did not receive enough money to build an active surveillance system and that as much as $50 million a year would be needed to do so.
The FDA official, who has long been familiar with the effort, said very little has come out of the project.
“It has been a huge waste of time and money,” the official said. “It was all in the service of industry.”
Public health experts and others said they worry that it’s only a matter of time before another emergency unfolds.
“Everybody at the FDA that I ever worked with — everybody — gets up in the morning and the one thing that they most worry about every day is, ‘Is there something on the market that’s going to hurt anybody?’ Nobody wants that on their heads,” said Larry Kessler, a former FDA official who spent 13 years at the agency before leaving in 2009. “When people’s lives are concerned, you want to take quicker action.”
“Government Is Supposed to Be There”

Long before his cancer diagnosis, former Louisiana Attorney General Ieyoub often talked to his family about good government. “Government is supposed to be there for people who don’t have a voice,” his son-in-law, Art Murray, recalled Ieyoub saying.
In recent years, the veteran prosecutor compared the Philips case to the battle he fought against tobacco companies in the 1990s. Even then, as the industry used cartoon characters to market cigarettes to children, Ieyoub fretted about the government’s failure to intercede.
“That’s the job of these agencies,” he said.
It’s one of the reasons he grew profoundly distressed after the Philips recall, his family members said.

Murray said Ieyoub believed the company was determined to keep the problem with its machines secret “and unfortunately a regulatory agency … fell right into that trap.”
“This is one of those perfect storm situations,” Murray said.
Ieyoub, who had recovered from mouth cancer, died of an aortic aneurysm in April. Known as “Giddie” to his five grandchildren, he was 78.



















